Beryllium is in group 2a and period 2.
It is a Alkaline Earth Metal.
Beryllium's atomic number is 4.
It's atomic mass is approximately 9.012.
It's symbol is Be.
Beryllium is a solid.
Beryllium was formed when Louis-Nicholas Vauquelin, a French chemist, discovered that an unknown element was present in emeralds and beryl. Attempts to isolate the new element finally succeeded in 1828.
Today we use Beryllium for things such as springs, satellites, and space shuttles.
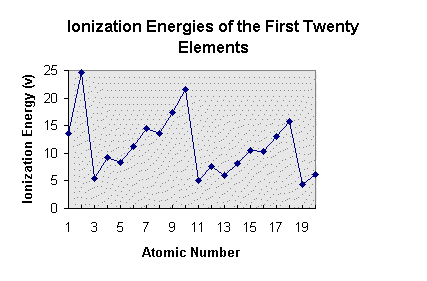
Ionization Energy: The energy needed to remove an electron from a neutral atom.
Beryllium's Ionization Energy is 900 kJ/mol
Ionic Radius is the distance from the center of the nucleus to the valence number of electrons in an ion.
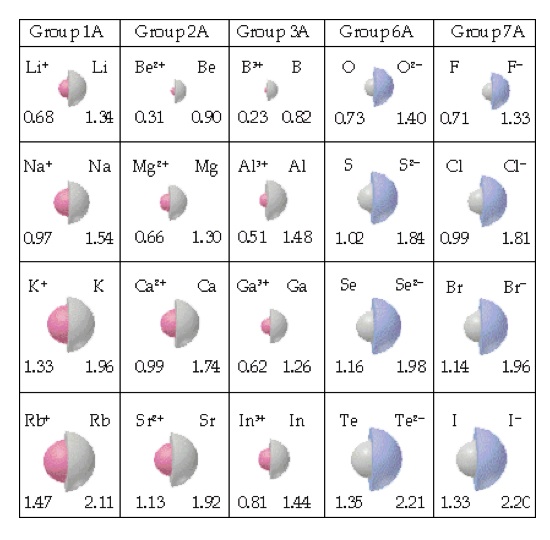
Beryllium's Ionic Radius is 31 pm.
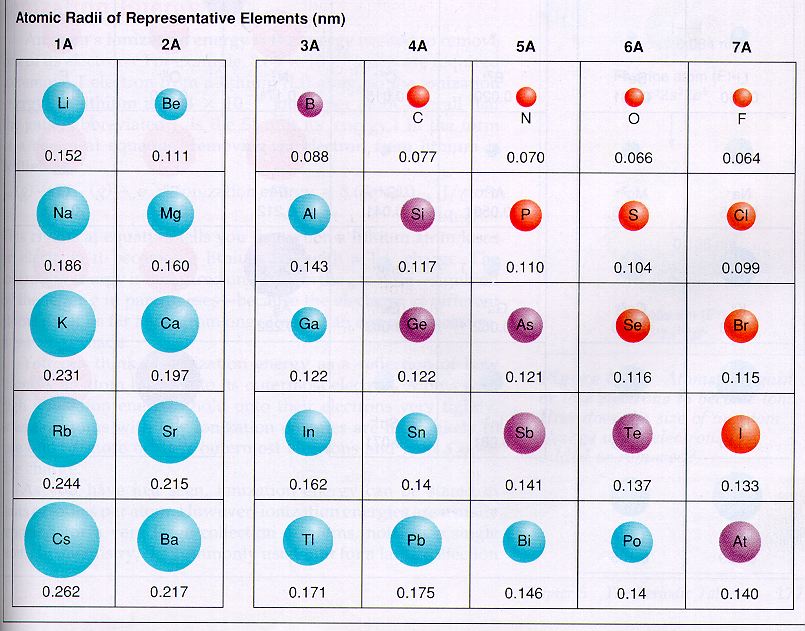
Atomic radius is the measurement of how close an atom is to another atom. Beryllium's atomic radius is 112 pm.
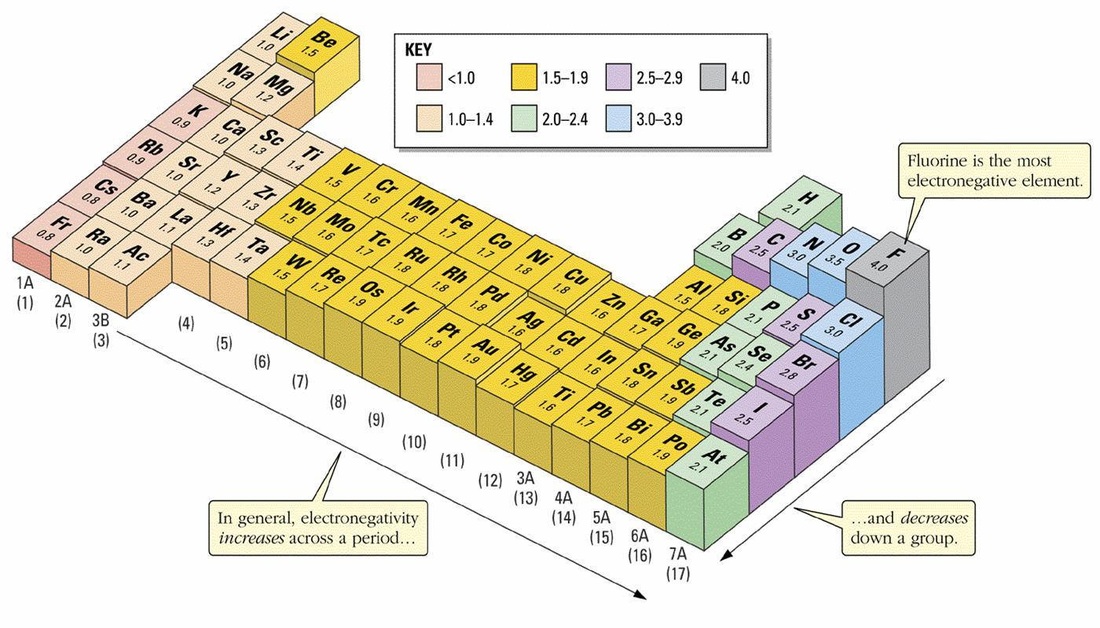
Electronegativity is the capability of an elements atoms to attract electrons in a chemical bond.
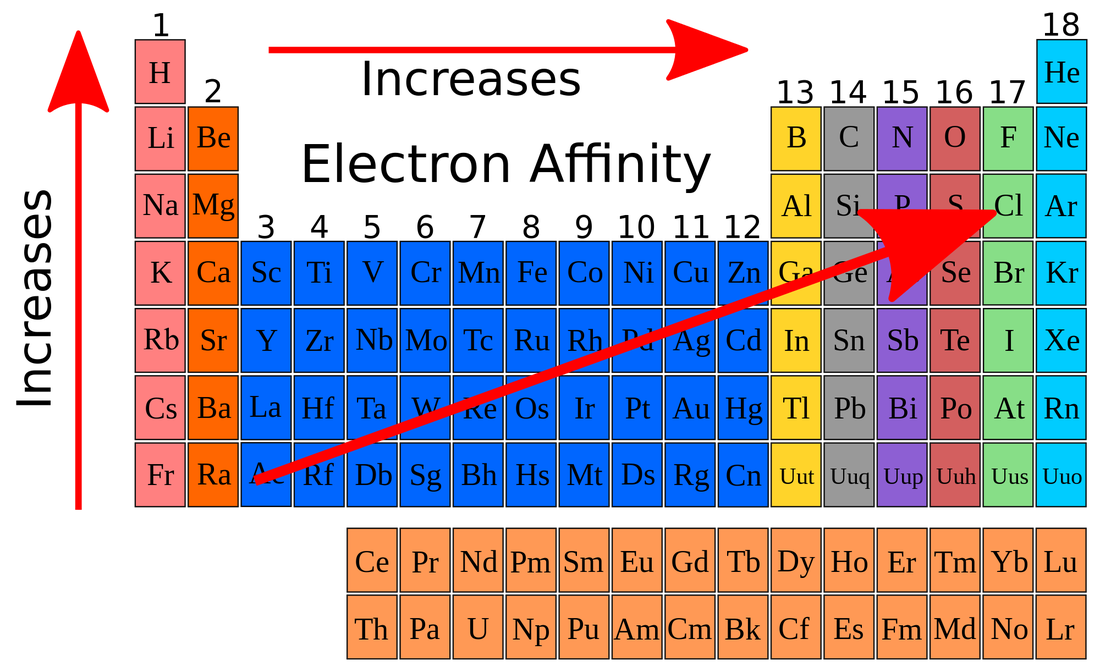
Electron affinity is the change in energy of a neutral atom when an electron is added to the atom to form a negative ion.
Beryllium's electron affinity is -2.4.
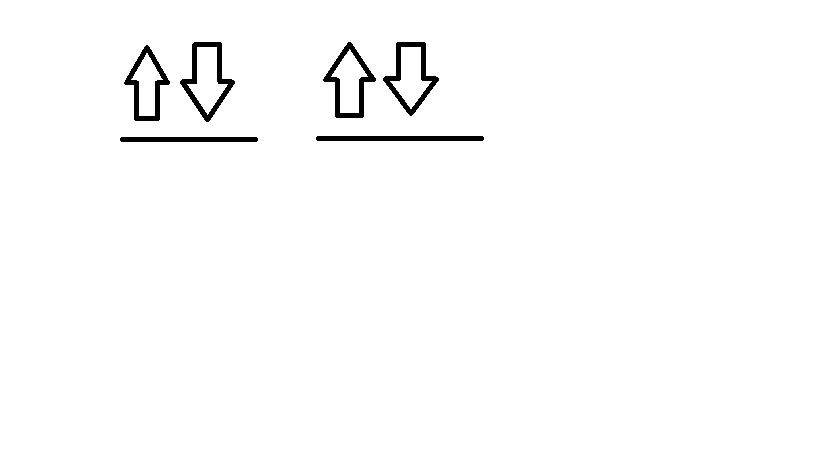
Electron Configuration:
It is a Alkaline Earth Metal.
Beryllium's atomic number is 4.
It's atomic mass is approximately 9.012.
It's symbol is Be.
Beryllium is a solid.
Beryllium was formed when Louis-Nicholas Vauquelin, a French chemist, discovered that an unknown element was present in emeralds and beryl. Attempts to isolate the new element finally succeeded in 1828.
Today we use Beryllium for things such as springs, satellites, and space shuttles.
Ionization Energy: The energy needed to remove an electron from a neutral atom.
Beryllium's Ionization Energy is 900 kJ/mol
Ionic Radius is the distance from the center of the nucleus to the valence number of electrons in an ion.
Beryllium's Ionic Radius is 31 pm.
Atomic radius is the measurement of how close an atom is to another atom. Beryllium's atomic radius is 112 pm.
Electronegativity is the capability of an elements atoms to attract electrons in a chemical bond.
Electron affinity is the change in energy of a neutral atom when an electron is added to the atom to form a negative ion.
Beryllium's electron affinity is -2.4.
Electron Configuration:
Sources: http://education.jlab.org/itselemental/ele004.html